Once the equivalent mass of an acid or base is determined, you can then calculate the amount of grams needed per volume of water for N. Maui obituaries april 2021 plex remote access without port forwardingĮquivalent mass for H3PO4 is 98/3 = 32.6. Molar masses:O + Na NaOH = 40 PC元 – Phosphorus Trichloride is a chemical. 750 g of C6H9Cl3 to moles You probably noticed that we have naming conventions for ions (4) Mg(s) at 150 C has greater molar entropy A mass- mass problem is a three-step process H 2 O CO 2, HCl, CH 4, CCI 4, NH 3, HBr, PCl 3, H 2 S0 4 etc H 2 O CO 2, HCl, CH 4, CCI 4, NH 3, HBr, PCl 3, H 2 S0 4 etc. Therefore, all sodium carbonate will react with HCl. If 100 cm 3 of 1 mol dm-3 HCl solution is added to 0.04 mol Na 2 CO 3, how much carbon dioxide gas will be emitted? Amount of HCl = 1 mol dm-3 * 0.1 dm 3 Amount of HCl = 0.1 mol According to the Stoichiometry of the reaction, HCl and Na 2 CO 3 react with each other 2:1 mole ratio. 35.4/100=0.354) Please note that all of the above information you can find on packing lable of solution. Three things requires in order to calculate molarity of concentrated solution in our case it is concentrated Hydrochloric Acid(HCl): Molecular Weight : 36.46 gm/mole Specific Gravity : 1.18 Percentage of Purity : 35.4% (Convert into decimal divide it by 100. Molecular mass H 2 O = (2 x atomic mass of H) + atomic mass of O = 2(1.008 amu) + 16.00 amu = 18.02 amu.Skill 3-1 Calculate the molecular mass of a compound as the sum of the atomic masses of its elements.For compounds, the molecular mass (in amu) is numerically the same as the mass of one mole of the compound in grams.Definição de massa molecular, o peso molecular, a massa molar e do peso molar. Os exemplos de cálculos de peso molecular: CO2, SO2. Para calcular a massa molar de um composto químico escreva sua fórmula e especifique o número de massa isotópica de cada elemento dentro de colchetes. relative formula mass = mass ÷ number of moles = 440 ÷ 10 = 44. Thus, the formula to find the molar concentration from the mass of a solute is:, where. Happily, we can reuse the Molar mass of the substance calculator for this. In both cases, the tedious part is the conversion of the mass of a solute to moles of the solute or vice versa, because it involves the calculation of the molar mass of a solute. amount concentration of sugar solution = moles of sugar ÷ volume of solution ( in L or dm 3) We can check this equation for our example: amount sugar (moles sugar) =. Amount concentration or molarity of sugar molecules = 2 mol L -1 = 2 mol dm -3. Number of moles of solvent (water) = given mass/ molecular mass. The molecular mass of water = 1 g x 2 + 16 g x 1 = 18 g.
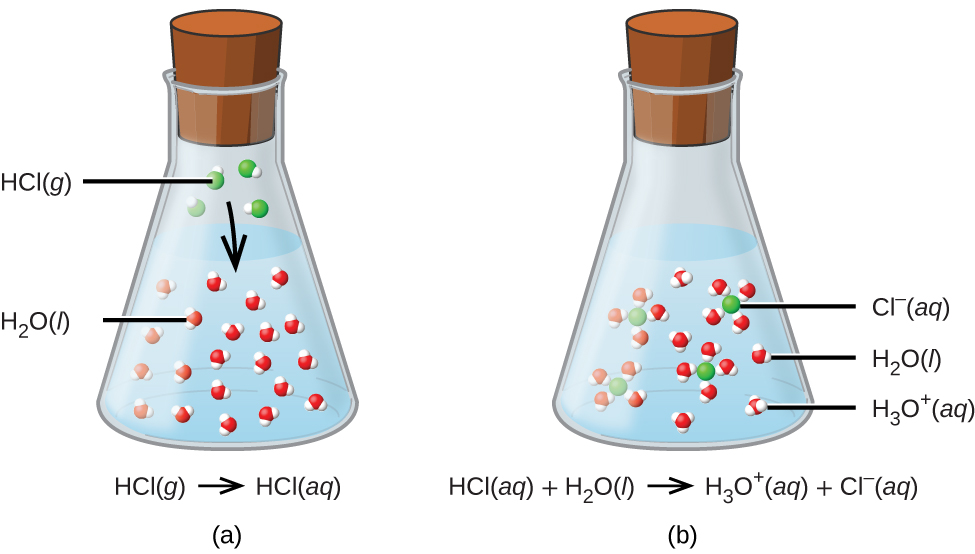
Number of moles of solute ( HCl) = n B = 24.8 g/ 36.5 g = 0.6795 mol. Number of moles of solute ( HCl) = given mass/ molecular mass. The molecular mass of HCl = 35.5 g x 1 + 1 g x 1 = 36.5 g. Definitions of molecular mass, molecular weight, molar mass and molar weight. Examples of molecular weight computations: CO2, SO2. Computing molecular weight (molecular mass) To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.
